CDM
Streamlining Data for Better Decision Making in Healthcare
Drug Development Stratagies
The right development strategy is critical for commercial success. With biotech companies investing in advanced therapies and international trials, there is an increased focus on regulatory planning, including the design of the clinical protocols, nonclinical testing plans and manufacturing processes. Hex info Technology provides first-class regulatory services to help expedite drug development within the complex and dynamic research environment.
Core Regulatory Services
Regulatory gap assessment and strategy
Design and review of nonclinical toxicology studies
Writing Investigator Brochures (IBs)
Global submissions including IND, DMF, NDA, BLA and combination products
Meetings and briefing packages including Type A, B and C meetings, scientific advice
Fast track and orphan designations
USA agent services
Institutional Biosafety Committee and GMO medicine applications
Design and review of nonclinical toxicology studies
Writing Investigator Brochures (IBs)
Global submissions including IND, DMF, NDA, BLA and combination products
Meetings and briefing packages including Type A, B and C meetings, scientific advice
Fast track and orphan designations
USA agent services
Institutional Biosafety Committee and GMO medicine applications
Core Medical Services
Medical writing
Clinical trial design
Medical monitoring
Pharmacovigilance
Clinical trial design
Medical monitoring
Pharmacovigilance
Liver Disease Trails
Hepatitis B (HBV)
Hepatitis C (HCV)
Hepatocellular carcinoma (HCC) and bile duct malignancies
Non-Alcoholic Steatohepatitis (NASH)
Non-Alcoholic Fatty Liver Disease (NAFLD)
Liver Transplant
Hepatitis C (HCV)
Hepatocellular carcinoma (HCC) and bile duct malignancies
Non-Alcoholic Steatohepatitis (NASH)
Non-Alcoholic Fatty Liver Disease (NAFLD)
Liver Transplant


Synopsis
Liver diseases are highly prevalent in the Asia-Pacific region. The region hosts some of the most active key opinion leaders and investigators with strong experience working with biotechnology companies on hepatology trials.
Script/Panellists
Arsalan Arif: Hi everyone, I am Arsalan Arif the publisher of Endpoints news, and I am pleased to be here today as your MC. We have got an important topic today, and a great panel. It is sponsored by and we will be talking about Asia-pacific as a global hub for hepatology clinical trials. Now I would like to introduce our panel.
First up we have Professor Man-Fung Yuen, Chief of the division of Gastroenterology-Hepatology, and the Deputy Head of the Department of Medicine at Queen Mary Hospital Hong Kong. Next, we have Dr. James Hamilton, Vice President and Head of Clinical Development at Arrowhead Pharmaceuticals. Next, we have Professor Ed Gane, Chief Hepatologist and Transplant Physician and Deputy Director of the New Zealand Liver Transplant Unit at Auckland City Hospital and then we have Professor Jacob George Head Department of Gastroenterology and Hepatology at Westmead Hospital. And our moderator is Barry Murphy, the Director of Business development .We've got a session here for you today, and if you've got questions you can clear it at the Q&A over there, but now I'm going to start the presentation over here and turn it over to Barry. Barry let us begin.
Barry Murphy: Thanks, Arsalan. So, okay we are very excited to have such a stellar line-up as he mentioned here today, and I also saw the registration list, and delighted that so many familiar names of people who work with both Novotech, […] and there are key opinion leaders with us here today. Very quickly for those who do not yet work with us, a quick intro and for a bit of context. So is a full-service Asia Pacific biotech focused CRO. And we've local teams on the ground in 11 countries across the region, so that’s Australia New Zealand where we started out, and then the big markets of China, Hong Kong, and Taiwan, and, as well as South Korea, and then Southeast Asia, and Singapore, Malaysia, Thailand, the Philippines, and then lastly India. So, our model has always been to hire and very experienced local, full-time regulatory, and clinical team on the ground in each country which we operate, and to really help our client base navigate the region in terms of local requirements; language, cultural nuances, etc. And we play a huge stock in our KOLs relationships […]. So, given the high incidence of liver related diseases in Asia Pacific, it really has become a core area of focus for us at Hexainfo Technology. Almost all of the Hepatitis work […] all has started with Professor Gane and his team at ACS, before expanding into Asia with Professor Yuen at Queen Mary, which is on the list there as one of the top sites. And across the region including ASAN, and Seoul National in South Korea; both sites […] we have memorandums of understanding in place with. So, it really is an area we work in quite a lot. In NASH, and PBC we have a lot of that as well, and we do a lot work with Professor George at Westmind and look forward to speaking on that side as well. So, […] exciting space now to have this line-up here with us today. On that note, I will first turnover to James from Arrowhead, a long-term client and James and a friend . I am very happy, and look forwards to your perspective on the region, and of the work you have done.
Barry Murphy: Thanks, Arsalan. So, okay we are very excited to have such a stellar line-up as he mentioned here today, and I also saw the registration list, and delighted that so many familiar names of people who work with both Novotech, […] and there are key opinion leaders with us here today. Very quickly for those who do not yet work with us, a quick intro and for a bit of context. So is a full-service Asia Pacific biotech focused CRO. And we've local teams on the ground in 11 countries across the region, so that’s Australia New Zealand where we started out, and then the big markets of China, Hong Kong, and Taiwan, and, as well as South Korea, and then Southeast Asia, and Singapore, Malaysia, Thailand, the Philippines, and then lastly India. So, our model has always been to hire and very experienced local, full-time regulatory, and clinical team on the ground in each country which we operate, and to really help our client base navigate the region in terms of local requirements; language, cultural nuances, etc. And we play a huge stock in our KOLs relationships […]. So, given the high incidence of liver related diseases in Asia Pacific, it really has become a core area of focus for us at Hexainfo Technology. Almost all of the Hepatitis work […] all has started with Professor Gane and his team at ACS, before expanding into Asia with Professor Yuen at Queen Mary, which is on the list there as one of the top sites. And across the region including ASAN, and Seoul National in South Korea; both sites […] we have memorandums of understanding in place with. So, it really is an area we work in quite a lot. In NASH, and PBC we have a lot of that as well, and we do a lot work with Professor George at Westmind and look forward to speaking on that side as well. So, […] exciting space now to have this line-up here with us today. On that note, I will first turnover to James from Arrowhead, a long-term client and James and a friend . I am very happy, and look forwards to your perspective on the region, and of the work you have done.
Medical Monitor
https://ccrps.org/clinical-research-blog/medical-monitor-responsibilities-in-a-clinical-trial
The Multifaceted Role of the Medical Monitor The complexities of clinical research in 2024 demand a multifaceted role for medical monitors.
Their responsibilities encompass:
Protocol Review and Design: Scrutinizing protocols to prioritize patient safety and ensure alignment with best practices. Consider our CRA course for detailed insights into this role.
Safety Oversight: Continuously monitoring patient well-being throughout the trial and addressing any safety concerns.
Data Analysis and Interpretation: Collaborating with the research team to analyze and interpret trial data effectively.
Regulatory Compliance: Ensuring the trial adheres to all relevant regulatory requirements.
Communication and Collaboration: Acting as a bridge between sponsors, investigators, and research teams, fostering clear communication and collaboration.
Medical Monitor vs. Principal Investigator (PI): PIs lead trials at a specific site, while medical monitors offer broader oversight across multiple trials and sites.
Medical Monitor vs. Clinical Research Associate (CRA): CRAs focus on protocol adherence and data collection at a single site, whereas medical monitors provide physician-level expertise for broader trial aspects.
The Multifaceted Role of the Medical Monitor The complexities of clinical research in 2024 demand a multifaceted role for medical monitors.
Their responsibilities encompass:
Protocol Review and Design: Scrutinizing protocols to prioritize patient safety and ensure alignment with best practices. Consider our CRA course for detailed insights into this role.
Safety Oversight: Continuously monitoring patient well-being throughout the trial and addressing any safety concerns.
Data Analysis and Interpretation: Collaborating with the research team to analyze and interpret trial data effectively.
Regulatory Compliance: Ensuring the trial adheres to all relevant regulatory requirements.
Communication and Collaboration: Acting as a bridge between sponsors, investigators, and research teams, fostering clear communication and collaboration.
Medical Monitor vs. Principal Investigator (PI): PIs lead trials at a specific site, while medical monitors offer broader oversight across multiple trials and sites.
Medical Monitor vs. Clinical Research Associate (CRA): CRAs focus on protocol adherence and data collection at a single site, whereas medical monitors provide physician-level expertise for broader trial aspects.
Understanding Medical Monitoring According to E6-GCP
The E6 Good Clinical Practice (GCP) guidelines define a medical monitor as a person responsible for supervising the clinical trial process. They ensure that protocols, standard operating procedures (SOPs), Good Manufacturing Practices (GMP), and regulatory requirements are all followed according to established standards as outlined in the European Medicines Agency's (EMA) ICH Guideline E6(R2) on Good Clinical Practice https://www.ema.europa.eu/en/human-regulatory-overview/research-and-development/compliance-research-and-development/good-clinical-practice
Alternative Terminologies for Medical Monitor
It's important to note that medical monitor is not the only term used for this role.
Here are some alternatives:.
Clinical research associate (CRA).
Site manager.
Senior CRA.
Clinical trial assistant (CTA).
However, clinical research associate (CRA) is the most frequently used alternative. Be aware that while there may be some overlap in responsibilities, CRAs
Here are some alternatives:.
Clinical research associate (CRA).
Site manager.
Senior CRA.
Clinical trial assistant (CTA).
However, clinical research associate (CRA) is the most frequently used alternative. Be aware that while there may be some overlap in responsibilities, CRAs
Difference between a Medical Monitor and Principal Investigator
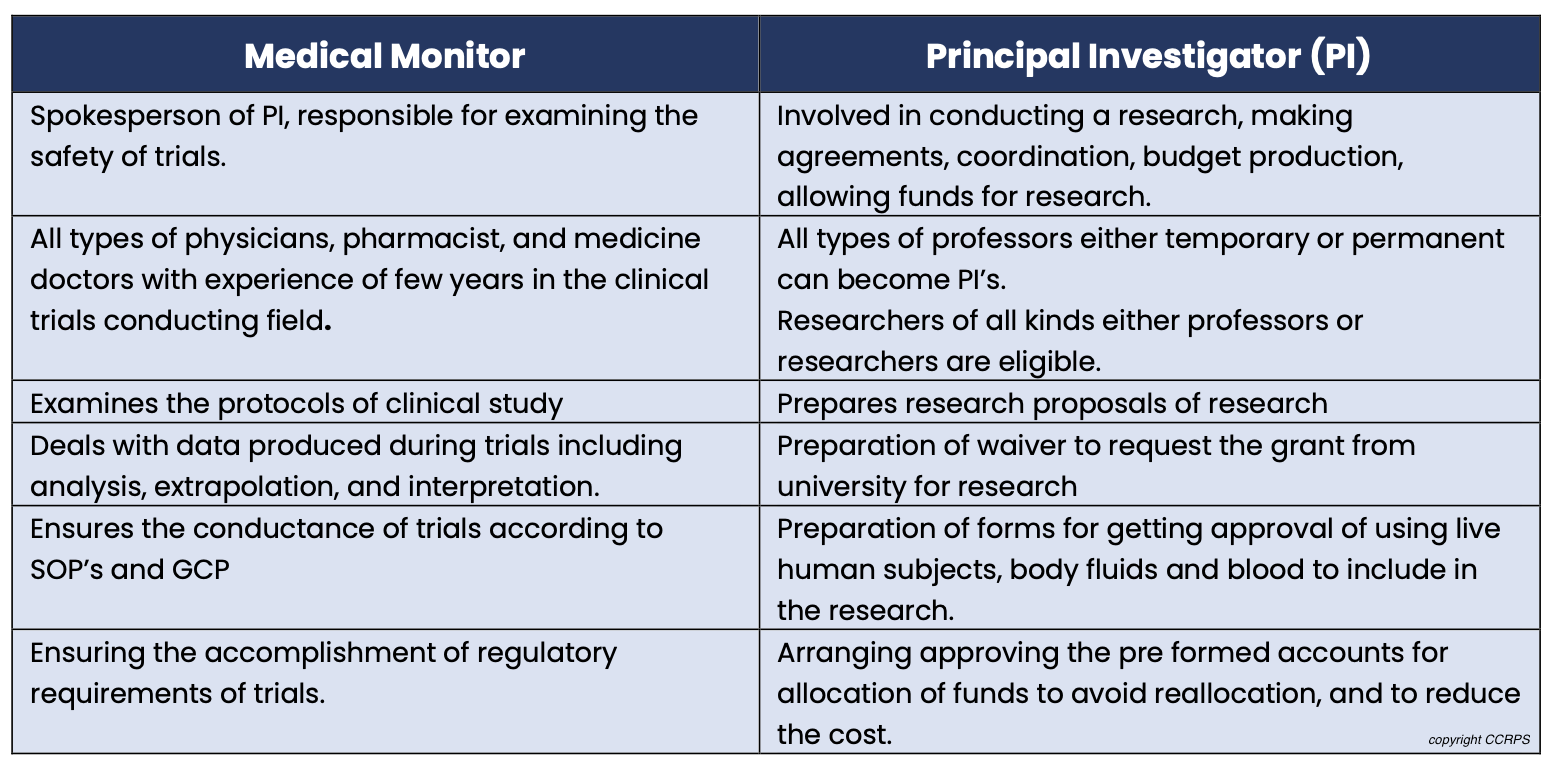
Skills and qualifications required to be a medical monitor:
Medical degree with strong leadership skills
Direct experience in the pharmaceutical industry (i.e. Clinical Research Associate; preferred; although with CCRPS Medical Monitor Certification you are qualified to work as both a CRA and MM)
The preferred countries for the experience are US and EU
Knowledge of both local and Global regulatory requirements as well as the knowledge of local and Global GCP
Skillful in converting input into regulatory documentation.
Strong communication skills to deal with stakeholders either internal or external.
Strong writing and presentation skills
. Skillful to deal with audience
especially medical and scientific community.
Relevant work experience in medical monitoring and/or pharmacovigilance and/or drug safety experience in a CRO, pharmaceutical, or clinical trial environment required.
Must have appropriate understanding about the International conference on Harmonization (ICH), GCP guidelines.
Must have appropriate knowledge of research, clinical trials and clinical terminologies.
Having strong decision-making power to deal with the trials.
Proficiency in English (written and verbal) required.
Capable of building and maintaining the trust of clients.
You can showcase and gain the knowledge you need to work as a MM with CCRPS’s Medical Monitor certification which trains you to a Senior Monitor’s level of knowledge to allow for easy promotion in the field. You do need experience before getting promoted so we suggest getting an internship while studying for the course prior to applying for Medical Monitor or CRA jobs!
Individuals who should try to be a medical monitor include:
Typically this is a fully qualified physician (MD, MBBS, IMG, FMG)
Sometimes residency in the work-specific department or in internal medicine is helpful (whether international or US-completed) In the US it is also permitted to also use a PharmD (pharmacist) as a medical monitor
Direct experience in the pharmaceutical industry (i.e. Clinical Research Associate; preferred; although with CCRPS Medical Monitor Certification you are qualified to work as both a CRA and MM)
The preferred countries for the experience are US and EU
Knowledge of both local and Global regulatory requirements as well as the knowledge of local and Global GCP
Skillful in converting input into regulatory documentation.
Strong communication skills to deal with stakeholders either internal or external.
Strong writing and presentation skills
. Skillful to deal with audience
especially medical and scientific community.
Relevant work experience in medical monitoring and/or pharmacovigilance and/or drug safety experience in a CRO, pharmaceutical, or clinical trial environment required.
Must have appropriate understanding about the International conference on Harmonization (ICH), GCP guidelines.
Must have appropriate knowledge of research, clinical trials and clinical terminologies.
Having strong decision-making power to deal with the trials.
Proficiency in English (written and verbal) required.
Capable of building and maintaining the trust of clients.
You can showcase and gain the knowledge you need to work as a MM with CCRPS’s Medical Monitor certification which trains you to a Senior Monitor’s level of knowledge to allow for easy promotion in the field. You do need experience before getting promoted so we suggest getting an internship while studying for the course prior to applying for Medical Monitor or CRA jobs!
Individuals who should try to be a medical monitor include:
Typically this is a fully qualified physician (MD, MBBS, IMG, FMG)
Sometimes residency in the work-specific department or in internal medicine is helpful (whether international or US-completed) In the US it is also permitted to also use a PharmD (pharmacist) as a medical monitor
https://ccrps.org/clinical-research-blog/medical-monitor-responsibilities-in-a-clinical-trial
Pharmacovigilance:
A Complete Guide to Pharmacovigilance and Drug Safety Training Feb 16 What Is Pharmacovigilance? - Definition, Jobs, Salary, And Pharmacovigilance Certification Pharmacovigilance A Guide All About Pharmacovigilance

To increase your chances of getting hired or promoted, scroll below or enroll in our CCRPS Advanced Pharmacovigilance and Argus Safety Certification (APVASC)TM Course which covers drug safety and pharmacovigilance training
What is pharmacovigilance?
Pharmacovigilance Is The Study Of Two Primary Outcomes In The Pharmaceutical Industry: Safety And Efficacy. Essentially, it asks does a drug work and is it safe?
Pharmacovigilance is the process of monitoring the effects of drugs, both new and existing ones. This includes collecting data, analyzing it, and taking steps to prevent any negative effects. Pharmacovigilance must happen throughout the entire life cycle of a drug, from when it is first being developed to long after it has been released on the market.
What is the Aim Of Pharmacovigilance?
Pharmacovigilance is required through the entire life cycle of a drug – starting at the preclinical development stage and going right through to continued monitoring of drugs once they hit the market.
Pharmacovigilance includes collecting, analyzing, monitoring, and preventing adverse effects in new drugs and therapies. It can be broken down into three main sub-specialisms:
1.Surveillance: Surveillance is geared towards risk management and signal detection. Roles in this specialism focus analysis of drug safety information gathered from other professionals. Surveillance is responsible for creating development safety update reports (DSURs) for drugs in clinical research and periodic benefit-risk evaluation reports (PBRER) for drugs that are on the market.
2.Operations: Operations focus on collecting and recording information during preclinical development, early clinical trials, and gathering real-world evidence (RWE) of adverse events reported by medical professionals and patients. Operations may also create standard operating procedures (SOPs), individual case study reports, and regulatory reports.
3.Systems: Systems is concerned with the development of robust systems to store and manage data relating to pharmacovigilance. It involves keeping abreast of changing regulations and guidance in the pharmacovigilance industry and ensuring compliance at all levels of an organization. The Qualified Person for Pharmacovigilance (QPPV) is responsible for ensuring that an organization's pharmacovigilance system meets all applicable requirements. Learn more
What is Pharmacovigilance Training?
If you’re looking to boost your career prospects in the pharmaceutical industry, drug safety training is a great string to add to your bow. Whether you want to move into clinical research or enhance your profile in your existing company, certification is crucial.
If you run a company and want to provide your staff with drug safety training to increase their knowledge and provide a safer working environment, our course is for you too.
We even help train Senior PVs from Fortune 500 companies to improve their efficiency and compliance.
No experience? No worries!
Our triple-accredited Advanced Pharmacovigilance and Argus Safety Certification (APVASC)TM is designed to teach you all you need to know in just 1 week!
Recognized Drug Safety Training I 180 Hours I On-Demand I Accredited I 25+ Modules I Training Compliance I Instant Enrolment I 1+Wk Certification I
What do Pharmacovigilance Officers do?
View fullsize
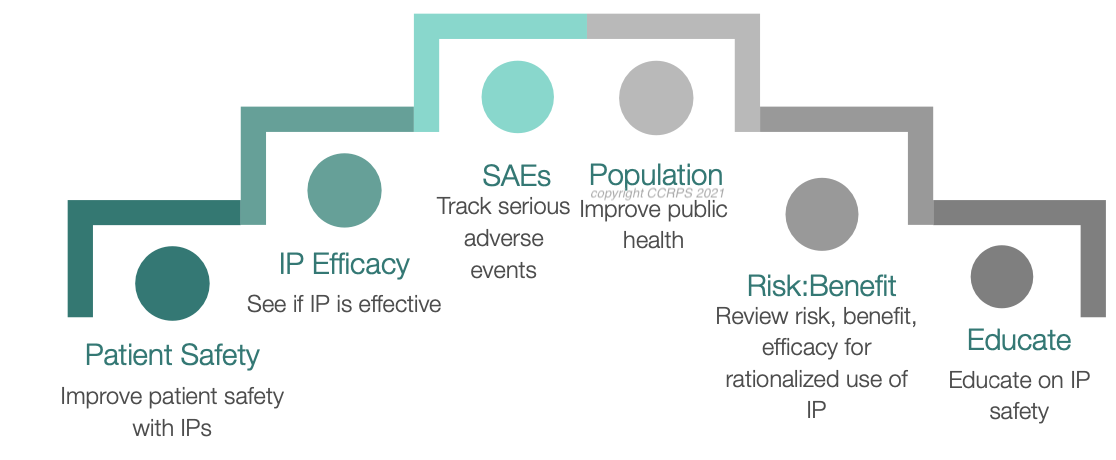
The exact nature of each role varies, but in essence, Pharmacovigilance Officers (PVs) collect adverse event data on drugs (Phase 4) to analyse and create usage warnings for the drug.
Some roles insist on physicians, nurses, or those with a Master of Science degree. A Master’s in pharmacovigilance is your best route into the industry – but that takes up to 2 years and is very expensive.
Your quicker route into the industry is with a drug safety accreditation.
Please note: A minimum of an associate degree is required to take any training course, including ours.
A Complete Guide to Pharmacovigilance and Drug Safety Training Feb 16 What Is Pharmacovigilance? - Definition, Jobs, Salary, And Pharmacovigilance Certification Pharmacovigilance A Guide All About Pharmacovigilance

To increase your chances of getting hired or promoted, scroll below or enroll in our CCRPS Advanced Pharmacovigilance and Argus Safety Certification (APVASC)TM Course which covers drug safety and pharmacovigilance training
What is pharmacovigilance?
Pharmacovigilance Is The Study Of Two Primary Outcomes In The Pharmaceutical Industry: Safety And Efficacy. Essentially, it asks does a drug work and is it safe?
Pharmacovigilance is the process of monitoring the effects of drugs, both new and existing ones. This includes collecting data, analyzing it, and taking steps to prevent any negative effects. Pharmacovigilance must happen throughout the entire life cycle of a drug, from when it is first being developed to long after it has been released on the market.
What is the Aim Of Pharmacovigilance?
Pharmacovigilance is required through the entire life cycle of a drug – starting at the preclinical development stage and going right through to continued monitoring of drugs once they hit the market.
Pharmacovigilance includes collecting, analyzing, monitoring, and preventing adverse effects in new drugs and therapies. It can be broken down into three main sub-specialisms:
1.Surveillance: Surveillance is geared towards risk management and signal detection. Roles in this specialism focus analysis of drug safety information gathered from other professionals. Surveillance is responsible for creating development safety update reports (DSURs) for drugs in clinical research and periodic benefit-risk evaluation reports (PBRER) for drugs that are on the market.
2.Operations: Operations focus on collecting and recording information during preclinical development, early clinical trials, and gathering real-world evidence (RWE) of adverse events reported by medical professionals and patients. Operations may also create standard operating procedures (SOPs), individual case study reports, and regulatory reports.
3.Systems: Systems is concerned with the development of robust systems to store and manage data relating to pharmacovigilance. It involves keeping abreast of changing regulations and guidance in the pharmacovigilance industry and ensuring compliance at all levels of an organization. The Qualified Person for Pharmacovigilance (QPPV) is responsible for ensuring that an organization's pharmacovigilance system meets all applicable requirements. Learn more
What is Pharmacovigilance Training?
If you’re looking to boost your career prospects in the pharmaceutical industry, drug safety training is a great string to add to your bow. Whether you want to move into clinical research or enhance your profile in your existing company, certification is crucial.
If you run a company and want to provide your staff with drug safety training to increase their knowledge and provide a safer working environment, our course is for you too.
We even help train Senior PVs from Fortune 500 companies to improve their efficiency and compliance.
No experience? No worries!
Our triple-accredited Advanced Pharmacovigilance and Argus Safety Certification (APVASC)TM is designed to teach you all you need to know in just 1 week!
Recognized Drug Safety Training I 180 Hours I On-Demand I Accredited I 25+ Modules I Training Compliance I Instant Enrolment I 1+Wk Certification I
What do Pharmacovigilance Officers do?
View fullsize

The exact nature of each role varies, but in essence, Pharmacovigilance Officers (PVs) collect adverse event data on drugs (Phase 4) to analyse and create usage warnings for the drug.
Some roles insist on physicians, nurses, or those with a Master of Science degree. A Master’s in pharmacovigilance is your best route into the industry – but that takes up to 2 years and is very expensive.
Your quicker route into the industry is with a drug safety accreditation.
Please note: A minimum of an associate degree is required to take any training course, including ours.
How To Get Into Pharmacovigilance
 ,
,The CCRPS pharmacovigilance certification to provide advanced training for entry level pharmacovigilance to ensure you are fully prepared for a career in drug safety monitoring. We are a non-profit organization dedicated to providing advanced pharmacovigilance training to students at all stages of their careers.
The courses provide you with theory and practical-based learning in pharmacovigilance and give you vital industry experience.
Our pharmacovigilance training courses meet
WHO-ISoP and FDA guidelines and are accredited by the ACCRE and other recognized accreditation boards.
Our Testimonials
Peoples Talk About Consilox

Christine Eve
Founder & CEO
The Consilox agency company also impressed us with their transpa regarding costs. The initial quote was best.

Kevin Smith
Customer
The Consilox agency company also impressed us with their transpa regarding costs. The initial quote was best.

Jessica Brown
Founder & CEO
The Consilox agency company also impressed us with their transpa regarding costs. The initial quote was best.

David Anderson
Customer
The Consilox agency company also impressed us with their transpa regarding costs. The initial quote was best.

Susan Neill
Founder & CEO
The Consilox agency company also impressed us with their transpa regarding costs. The initial quote was best.






